The concept of gene therapy, which originated in the late 1960s, has come a long way - having had its own share of significant setbacks and gargantuan success.
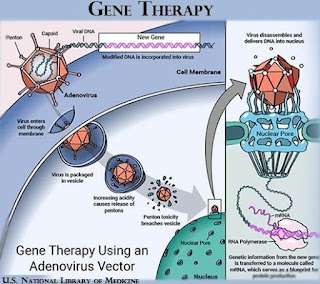
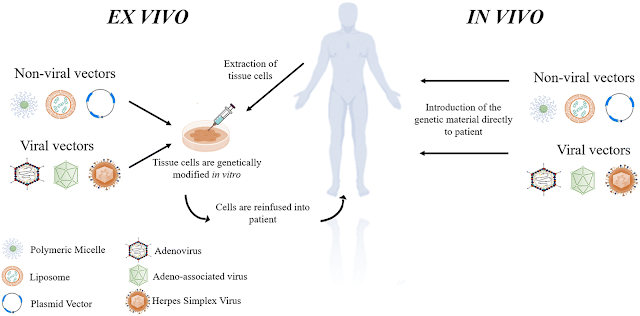
Gene therapy is a technique that uses genes to treat or prevent diseases caused by defective or missing genes. This approach helps to address the underlying cause of the disease, rather than offering solutions that focus only on the disease symptoms.
CRISPR the technology — not to be confused with Crispr Therapeutics, the company — builds on a project that sequenced the human genome. The first map cost $2.7 billion and was completed in 2003. Since then, the cost to map an individual's genome has dropped precipitously and could come down to just hundreds of dollars in the next few years.
The world's first gene therapy, Gendicine, was approved in China in 2004. Developed by SiBiono GeneTech Co., Ltd., Gendicine is indicated for head and neck squamous cell carcinoma. A year later, i.e., in 2005, another gene therapy product - Oncorine - was launched in China, by Shanghai Sunway Biotech Co. Ltd., for the treatment of head and neck cancer.
Russia launched its first gene therapy, Neovasculgen, to treat Peripheral Arterial Disease in 2011.
The first gene therapy approved by the European Commission was Glybera. Developed by uniQure N.V. (QURE), Glybera was approved in October 2012 for adult patients diagnosed with familial lipoprotein lipase deficiency and was launched in early 2015. However, the drug was withdrawn from the European Union in 2017 due to commercial failure. The second gene therapy to be greenlighted in the EU is Strimvelis, which was approved in 2016, for a rare disease called severe combined immunodeficiency due to adenosine deaminase deficiency, or ADA-SCID, dubbed "Bubble Boy" disease.
In the U.S., the first-ever gene therapy to win the regulatory nod is Novartis' (NVS) Kymriah. Approved by the FDA on August 30, 2017, Kymriah is indicated for Acute Lymphoblastic Leukemia, and Large B-Cell Lymphoma.
2017 saw two more gene therapies get the FDA nod - Kite/Gilead's (GILD) Yescarta, approved in October 2017, for the treatment of adults with diffuse large B-cell lymphoma, and Spark Therapeutics' Luxturna, approved in December 2017, to treat children and adult patients with an inherited form of vision loss that may result in blindness.

No comments:
Post a Comment